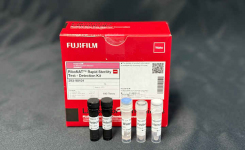
We have developed an improved Monocyte Activation Test (MAT) for pyrogen testing. The MAT serves as the replacement for the Rabbit Pyrogen Test and can be used to test parenteral drugs for endotoxin and non-endotoxin contaminations. The MAT kit includes all the required consumables to perform the Monocyte Activation Test, making it convenient to perform. In addition to the kit we provide consulting services and technical support in setting up the MAT in your organisation.
Microbiology Product Areas
LATEST MICROBIOLOGY NEWS
-
Real-Time Monitoring for Faster QC Microbiology
03 Feb 2026 -
Nipah Virus Antigens and Antibodies
03 Feb 2026 -
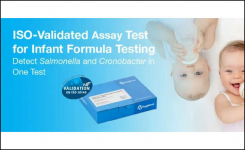
foodProof Salmonella Cronobacter
29 Jan 2026
MICROBIOLOGY EVENTS
-
WHX Labs Dubai
9 Feb 2026 -
Optimize Your Laboratory’s Pathogen Workflow
10 Feb 2026 -
Webinar: Advancing 3-day Sterility Testing for Cell and Gene Therapies
11 Feb 2026 -
Legionella Enumeration by ISO 11731: Limits and Biases
16 Feb 2026 -
Allergen Management Solutions for Food Manufacturing Webinar
18 Feb 2026 -
World Water-tech Innovation Summit
24 Feb 2026 -
4th International Beverages Quality Conference 2026
3 Mar 2026 -
Forum LABO 2026
10 Mar 2026 -
2026 QC Micro Summit
17 Mar 2026 -
Advanced Therapies
17 Mar 2026